
The Paul-Ehrlich-Institut strongly appeals to all individuals to take the opportunity to get vaccinated now, because COVID-19 vaccination offers the best protection against infection and disease. The available data, including data on safety of the vaccines, has quickly become more comprehensive than ever before for a newly introduced vaccine. Several billion people worldwide have now been vaccinated against COVID-19. In addition, vaccinations reduce the spread of the coronavirus. COVID-19 vaccines have prevented many people from becoming severely ill with COVID-19 or even dying. The vaccination campaign in Germany has been running for more than a year now. 
Given the fact that vaccination protection after primary immunisation with one of the COVID-19 vaccines is lower against the widespread Omicron coronavirus variant, booster vaccination is of great importance. The highest possible vaccination rate is desirable to protect the population from COVID-19 and from severe courses of the disease in particular. Two vaccine doses are to be given at least 3 weeks apart in order to achieve primary immunisation. The Standing Committee on Vaccination ( Ständige Impfkommission, STIKO) at the Robert Koch-Institut recommends Nuvaxovid for primary immunisation of people aged 18 and over. This protein-based COVID-19 vaccine showed comparable efficacy to the mRNA vaccines against COVID-19 in clinical trials during the marketing authorisation process. 
Instead, Nuvaxovid contains a genetically engineered spike protein of the coronavirus SARS-CoV-2, supplemented by the Matrix-M adjuvant. the spike protein blueprint, being inoculated. With protein-based vaccines, there is no genetic information, i.e. The authorisation of Nuvaxovid means that a protein-based vaccine has been added to the group. 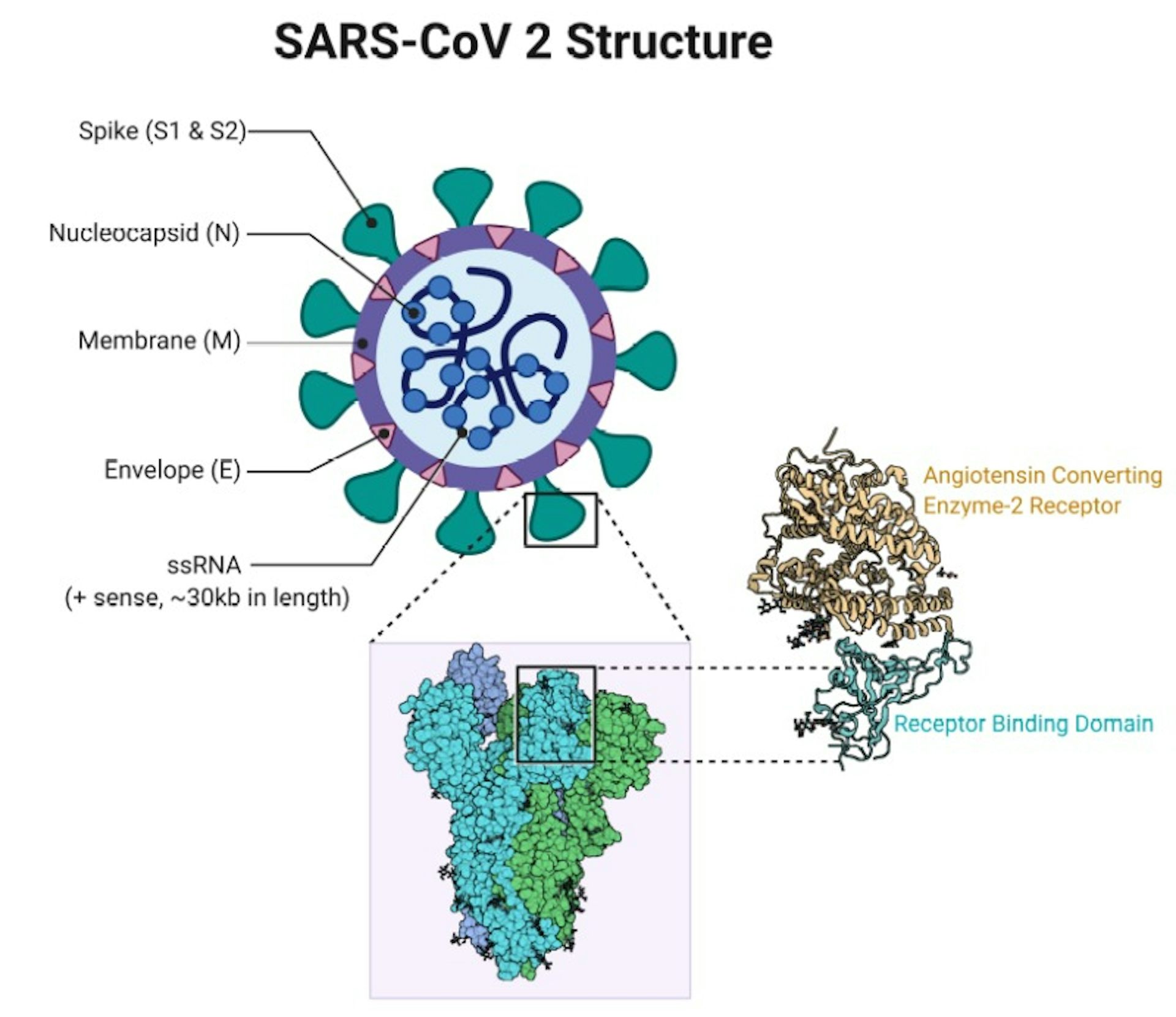
A conventional vaccine is now available to those who have not yet decided to get a COVID-19 vaccination. Two vector and two mRNA vaccines against COVID-19 have been available thus far in Germany. The federal government and the federal states are currently coordinating the exact modalities of distribution. At present, an additional agreement has been reached with the company Novavax for further deliveries of up to 30 million doses for Germany for the months of April to June 2022. This is to be followed by 900,000 doses in each of the following two weeks and 600,000 doses in the week beginning 14 March 2022. As things stand, the first partial delivery of an initial 1.4 million vaccine doses will be made to the German federal states in calendar week 8/2022. According to the EU Commission, the first deliveries are to arrive over several phases in February and March. A shipment of about four million vaccine doses is scheduled to be delivered to Germany in the first quarter. Germany will receive up to 34 million total vaccine doses of Nuvaxovid in 2022, according to the Federal Ministry of Health ( Bundesgesundheitsministerium, BMG). This COVID-19 vaccine, which received its European marketing authorisation on 20 December 2021, is the first protein-based COVID-19 vaccine to be used in Europe. The Paul-Ehrlich-Institut started the first batch release for the Nuvaxovid vaccine from the US pharmaceutical company Novavax on 22 February 2022 in Germany.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
